What’s the Science Behind Genomics?
This blog post is the second in a two-part series exploring the disruptive impact of genomics on healthcare and medicine. To read the first part, which focuses on the catalysts supporting genomics’ emergence as a powerful theme, click here.
There are several segments within the genomics field that stand to benefit from falling genetic testing costs, the rise of precision medicine ever-increasing amounts of genetic data, and other trends fuelling genomics’ disruption of health care. This piece offers a deep-dive into the specific technologies and scientific practices shaping the growth of this rapidly emerging industry, including:
- Genome sequencing
- Computational genomics and genetic diagnostics
- Development and testing of genetic medicines and therapies
- Gene editing
- Biotechnology
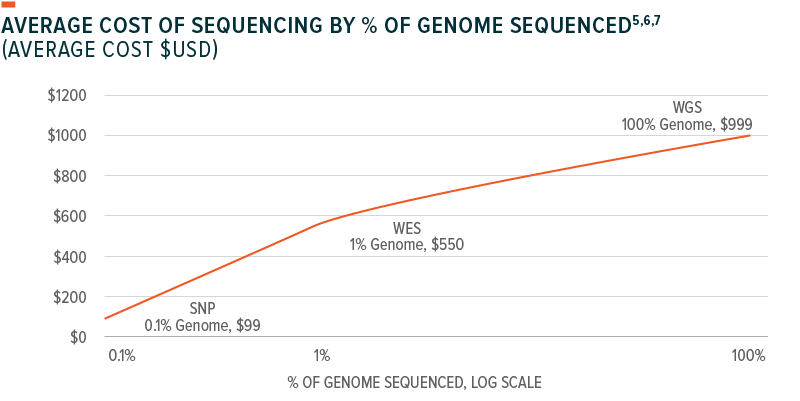
Genome sequencing: is the process of determining the exact order of one’s DNA. Estimates suggest that the global DNA sequencing market was valued at US$6.2 billion in 2017 and is expected to reach US$25.5 billion in 2025, with a compound annual growth rate (CAGR) of 19%.1 There are currently three common sequencing types available: SNP genotyping, whole exome sequencing (WES), and whole genome sequencing (WGS).
By sequencing a small, yet significant portion of one’s DNA, SNP genotyping checks for common genetic variants at a reduced cost. Popular genetic testing companies like 23andMe, use SNP genotyping for this reason. Similarly, by focusing on protein-encoding regions, WES analyses key gene expression pathways without coding the entire genome. After all, modern humans share up to 99% of their DNA with chimpanzees, meaning that mapping the whole genome for each person may not be the most efficient approach.3
WGS offers the most comprehensive insights, and while generally 10x more expensive than SNP genotyping, new practices such as multiplexing (i.e., analysing millions of DNA slices simultaneously), are making WGS cheaper and more efficient than before. New pocket-sized machines will also empower doctors and scientists to conduct sequencing in real time in the field to quickly generate actionable insights. Regardless of the method, however, both consumers and healthcare industry participants are driving sequencing demand in hopes of finding new ways to promote longer and healthier living.4
Computational genomics and genetic diagnostics: uses computational and statistical analysis to decipher biological insights from genome sequences and related data. The global computational biology market is expected to value US$6.8 billion in 2024, up from US$2.3 billion today with a CAGR of 19.5%.8 Among the primary benefits of computational genomics is improved understanding of gene expression, protein signalling pathways and their role in complex diseases such as diabetes, cardiovascular disease, Alzheimer’s and cancer. With increased volume of genetic data, data sharing, and improved reliability and validity of statistical models, computational genomics should help identify new treatment entry points and improve understanding of individual risk factors. For example, researchers are “letting loose” machine learning algorithms and neural networks on patient data to identify the underlying causes of addiction or dementia, or determining what genes, or combination of genes, fuel disease progression. In addition to the focus on humans, gene analytics have massive implications for other industries like agriculture and food & beverage. In agriculture for instance, computational genomics could improve crop diversity and resistance to climate change and other environmental pressures. Restaurants may even one day soon begin customising dining experiences based on your ‘taste’ genes.

Development and testing of genetic medicines and therapies: seeks to detect, cure or treat diseases by identifying and/or modifying an organism’s gene expression or functioning. Gene expression is an important concept here because it describes the processes by which DNA dictates our biological functions, often through protein signalling pathways. One objective of gene therapy, for example, might be the inactivation of a gene mutation by manufacturing an appropriate “blocker” that impedes a certain part of the gene expression pathway. Another type might add functional genes to compensate for defective genes or create a much-needed protein. Several gene therapies have already been approved by the FDA, the first of which was LUXTARNA in 2017, used to treat inherited blindness. Gene therapies are commonly delivered via a bone marrow transplant. Estimates suggest the gene therapy market should grow to US$4.4 billion by 2023, up from just US$584 million in 2016 with an impressive CAGR of 33.3%.9
Gene editing: inserts, deletes or replaces DNA at a specific site in the genome of an organism. While gene editing is often categorised as an extension of gene therapy, gene editing focuses on directly changing an individual’s DNA in the afflicted location, whereas gene therapy may use treatment options that affect gene expression pathways but do not alter DNA. Both types of solutions should complement each other given the diversity of genetic drivers of disease. Much of the excitement around gene editing surrounds CRISPR-Cas9 technology, which allows scientists to either slice open DNA at a specific location or substitute individual DNA units one-by-one without altering the entire strand. CRISPR-Cas9 editing has already been deemed to potentially address two/thirds of the more than 50,000 genetic diseases caused by a single gene.10 Other methods of gene editing involve another type of enzyme known as Zinc Finger Nucleases (ZFNs). While there are risks to both approaches, such as causing mutations in unintended locations, gene editing methods are improving.11 The first human CRISPR trial in the West just began in early 2019 to address a rare blood disorder, beta thalassemia. Similar efforts are anticipated to begin this year for other diseases like sickle cell anaemia, both in the United States and Europe. The entire healthcare industry will be watching closely to learn from the results of these early clinical trials and how they could potentially speed up opportunities to treat those afflicted with genetic disorders. Estimates suggest the gene editing market should reach US$7.5 billion by 2024, up from US$3.0 billion in 2017 with a CAGR of 14.5%.12 This number is expected to grow significantly if the treatment methods tested in clinical trials receive positive results and regulatory approval.
Biotechnology: Given the foundational role of genes in human biology, there are many synergistic opportunities with other parts of the biotechnology industry. Stem cell research and regenerative medicine, combined with advances in genomics, could help improve the effectiveness of tissue and organ transplants, for example. Such work could also improve our understanding of aging by collaborating with telomere research. In any case, genomics companies are likely to operate in a broad biotechnology ecosystem with many external partners to conduct the best research, produce the most innovative treatments and usher in an era of personalised medicine.
The rise of genomics and precision medicine are expected to fundamentally change how health care is understood and delivered. Human biology’s rapid convergence with big data, advanced analytics and scalable computing power should offer unprecedented insight into how diseases of all kinds afflict mankind, and even more importantly, how they may be prevented. From the use of new therapies to altering our own DNA, genomics’ far-reaching potential to transform health care creates a compelling thematic investing opportunity.
This document is not intended to be, and does not constitute, investment research as defined by the Financial Conduct Authority
This document is not intended to be, or does not constitute, investment research
